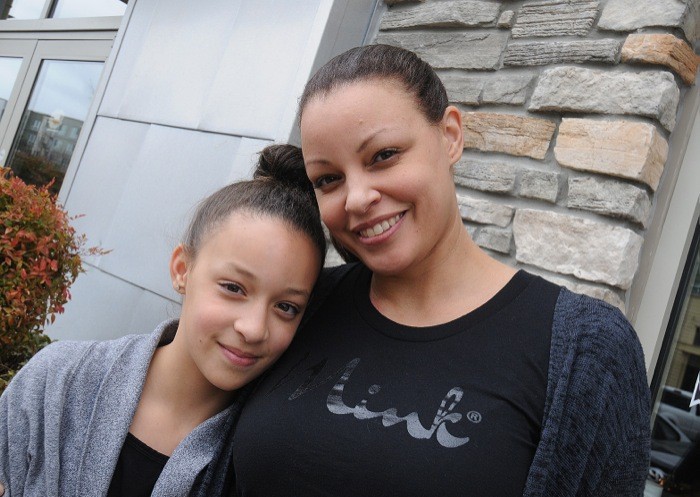After nine years fighting a drug company over her daughter’s heart defect, a Surrey mom is part of a group receiving a $6.2-million settlement.
During her pregnancy, Faith Gibson was taking Paxil, a drug for anxiety and depression manufactured by GlaxoSmithKline.
When her daughter Meah Bartram was born, she had a hole in her heart.
Meah was born in September of 2005, the same month that GlaxoSmithKline issued a warning that the drug might cause “congenital malformations… most commonly cardiovascular” in babies. The most common problem was a hole in the heart.
Paxil has been manufactured since the early 1990s. It is in a class of a drugs known as selective serotonin reuptake inhibitors (SSRIs).
Health Canada and its U.S. counterpart issued warnings about the potential hazard Paxil poses to the unborn.
Meah is now 11 years old and by all appearances is doing fine.
“She’s doing okay,” Gibson told The Leader in an interview Wednesday. “I guess the hard thing with stuff is anytime… she runs, or has a pain in her chest, or has cramps, she feels like there’s something wrong with her heart.”
There were times during her eight-year legal battle when Gibson felt discouraged, but she never gave up her resolve.
“The whole time I’m like, ‘no, I want to see this all the way through, I don’t care how long it takes’,” Gibson said.
As part of the settlement, GlaxoSmithKline agreed to pay $6.2 million to the fewer than 50 people in the class action lawsuit against the drug company.
Once the settlement is distributed, Gibson said she’ll put the money in a trust fund for her daughter.
Vancouver lawyer David Rosenberg told The Leader it was a tough battle, particularly getting class action certification, a move that was won and then appealed.
The case was scheduled for a full trial in January when the parties settled on the eve of proceedings getting underway.
“It was quite a complicated and difficult case,” Rosenberg said, adding one of the strongest parts of his case was the “very significant consequences” some of the new mothers and their babies faced.
“It’s heartbreaking,” he said.
GlaxoSmithKline (GSK) has not admitted to, nor has it been found, responsible for Paxil causing the heart defects.
“GSK does not admit to any liability or wrongdoing as part of the proposed settlement, but has agreed to resolve the class action to avoid the time and expense associated with the trial and the subsequent steps in the class action proceeding,” a representative from GlaxoSmithKline said in an email to The Leader.
GlaxoSmithKline said customer safety continues to be its highest aim.
“We continue to be of the view that the scientific evidence does not establish that exposure to Paxil during pregnancy causes cardiovascular birth defects,” the company spokesperson said. “Patient safety is our highest concern and we continue to believe that the company provided accurate and updated information in relation to Paxil to regulators, and communicated important safety information to regulatory agencies, the scientific community and healthcare professionals.”
Any women who took Paxil during their pregnancy and have a child with defects, particularly in the heart, are asked to email david@rosenberglaw.ca to share in the award.
The deadline to become part of the settlement is March 26 at 5 p.m.